The Most Delicious Smelling Experiment So Far

The last project saw me burning out an LED, this one has me slicing into lemons.
Mmmmm, lemons. (Michelle had no problem giving those a lick! But I bet it was more intense than the battery in project 1!)
This was the typical kitchen experiment of making a battery out of lemon halves, using zinc-coated nails and copper pennies as electrodes. I've actually done this experiment when I was young, but it was nice to have a refresher.
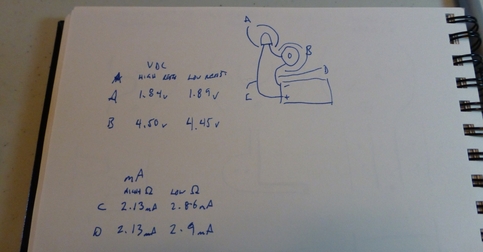
The first task was inserting the electrodes and probing with the multimeter: .9 volts! That was more than I was expecting. Again, I still don't really know what a volt is (and I forgot to measure the current at this stage) so I don't actually know if I should have been surprised or not. But I was.
The book didn't comment on which end of the lemon battery was the positive or negative, so I also took this opportunity to note which way around read positive voltage on the multimeter. (Spoiler: The copper terminal is the positive one.)
Mmmmm, lemons. (Michelle had no problem giving those a lick! But I bet it was more intense than the battery in project 1!)
This was the typical kitchen experiment of making a battery out of lemon halves, using zinc-coated nails and copper pennies as electrodes. I've actually done this experiment when I was young, but it was nice to have a refresher.
The first task was inserting the electrodes and probing with the multimeter: .9 volts! That was more than I was expecting. Again, I still don't really know what a volt is (and I forgot to measure the current at this stage) so I don't actually know if I should have been surprised or not. But I was.
The book didn't comment on which end of the lemon battery was the positive or negative, so I also took this opportunity to note which way around read positive voltage on the multimeter. (Spoiler: The copper terminal is the positive one.)

Wiring 4 lemon halves together in series produced 3.5 volts, and about 100 milliamps of current. (Side note: the rubber caps on these clips are kind of slippery and actually make it incredibly hard to squeeze open the jaws. The darn things always slip out from under your thumb and twist sideways.)
Finally, I wired in a small LED and it faintly but distinctly lit. It's interesting because it was pretty unremarkable. I remember when I did this experiment as a kid I was also unimpressed by the lit bulb or whatever. But seeing the numbers jump on the multimeter was exciting! Note if you are trying to make kitchen electronics interesting: Buy a $10 multimeter!
Apparently over time as the zinc is harvested for electrons, it will start oxidizing and turn blackish. As I type this I have the battery short-circuited in hopes of tinting the nails.
Aaaaaand that's the end of chapter 1! I'm really looking forward to the next chapter, as we start building circuits with functionality, and that's really why we're here, isn't it.
Finally, I wired in a small LED and it faintly but distinctly lit. It's interesting because it was pretty unremarkable. I remember when I did this experiment as a kid I was also unimpressed by the lit bulb or whatever. But seeing the numbers jump on the multimeter was exciting! Note if you are trying to make kitchen electronics interesting: Buy a $10 multimeter!
Apparently over time as the zinc is harvested for electrons, it will start oxidizing and turn blackish. As I type this I have the battery short-circuited in hopes of tinting the nails.
Aaaaaand that's the end of chapter 1! I'm really looking forward to the next chapter, as we start building circuits with functionality, and that's really why we're here, isn't it.
















 RSS Feed
RSS Feed